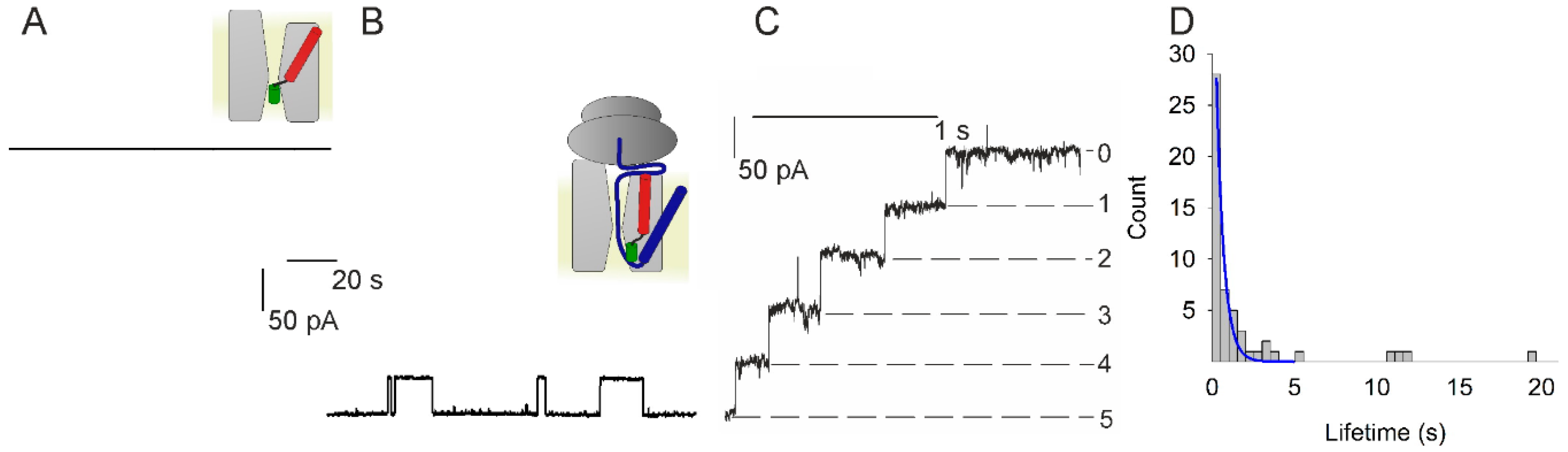
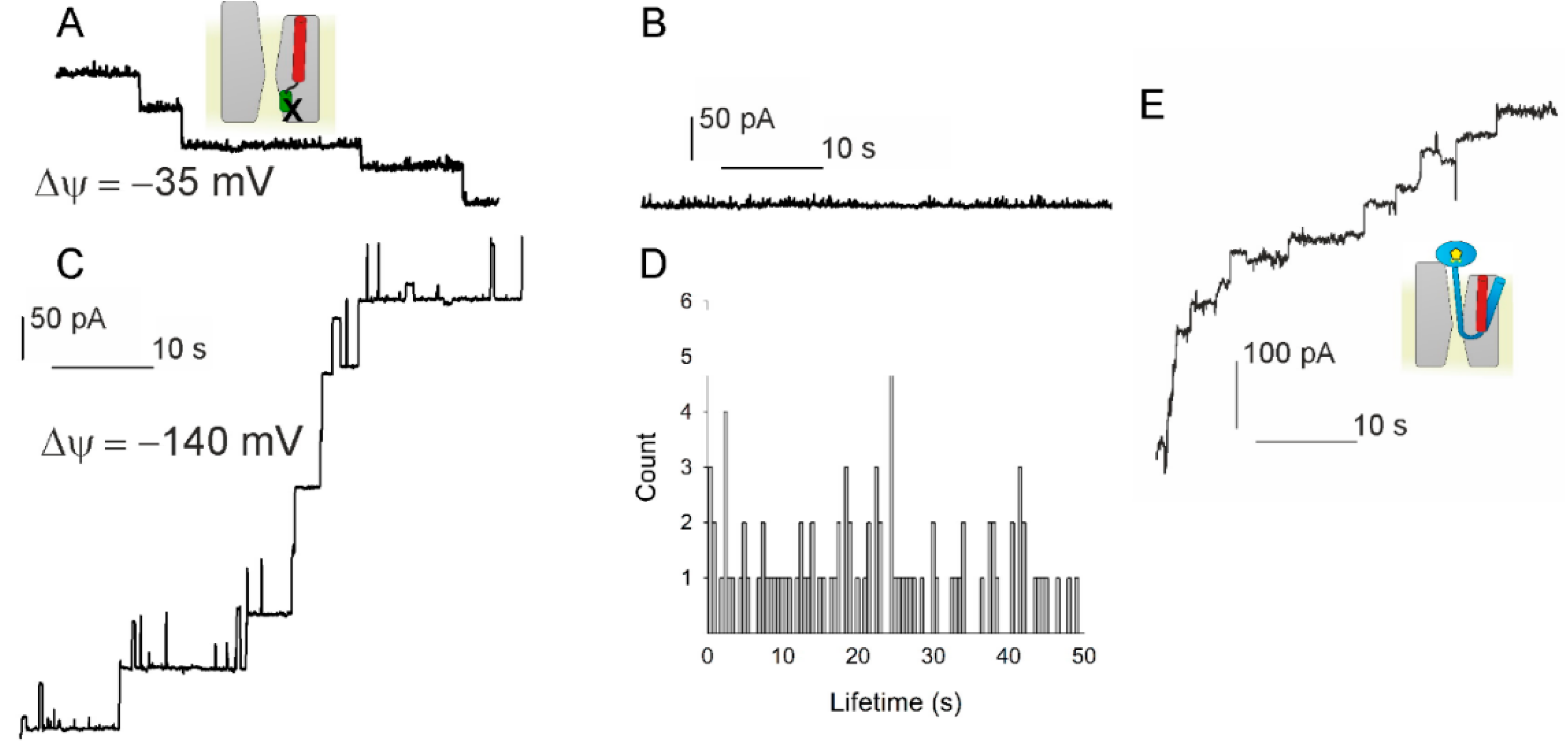
How guanylate-binding proteins achieve assembly-stimulated processive cleavage of GTP to GMP | Nature
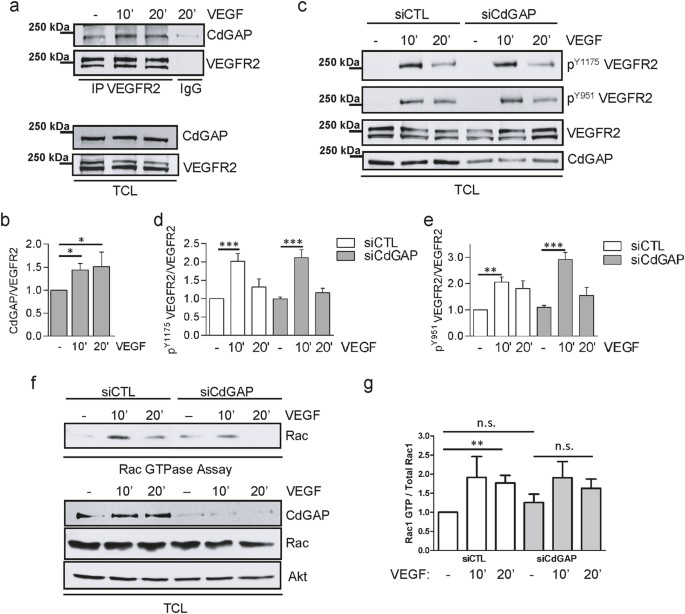
CdGAP/ARHGAP31, a Cdc42/Rac1 GTPase regulator, is critical for vascular development and VEGF-mediated angiogenesis | Scientific Reports

Role of Nucleotide Binding and GTPase Domain Dimerization in Dynamin-like Myxovirus Resistance Protein A for GTPase Activation and Antiviral Activity* - Journal of Biological Chemistry
Poster session abstracts – topic of research paper in Biological sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.

The Chaperone Protein SmgGDS Interacts with Small GTPases Entering the Prenylation Pathway by Recognizing the Last Amino Acid in the CAAX Motif* - Journal of Biological Chemistry

FKBP25/FKBP3 Protein Overview: Sequence, Structure, Function and Protein Interaction | Sino Biological

Adeno-Associated Virus Type 2-Mediated Gene Transfer: Role of Cellular FKBP52 Protein in Transgene Expression | Journal of Virology

GEF mechanism revealed by the structure of SmgGDS-558 and farnesylated RhoA complex and its implication for a chaperone mechanism | PNAS
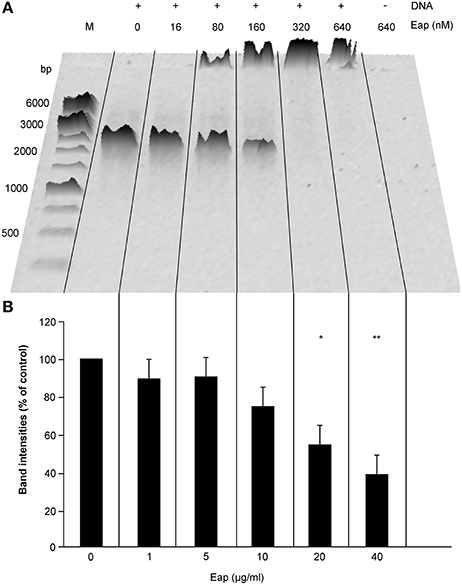
Frontiers | The Staphylococcus aureus Extracellular Adherence Protein Eap Is a DNA Binding Protein Capable of Blocking Neutrophil Extracellular Trap Formation | Cellular and Infection Microbiology

Protein disulfide isomerase externalization in endothelial cells follows classical and unconventional routes - ScienceDirect

Inhibition and Termination of Physiological Responses by GTPase Activating Proteins | Physiological Reviews