PDF) Aqueous Solutions of Cr(III) Sulfate: Modeling of Equilibrium Composition and Physicochemical Properties

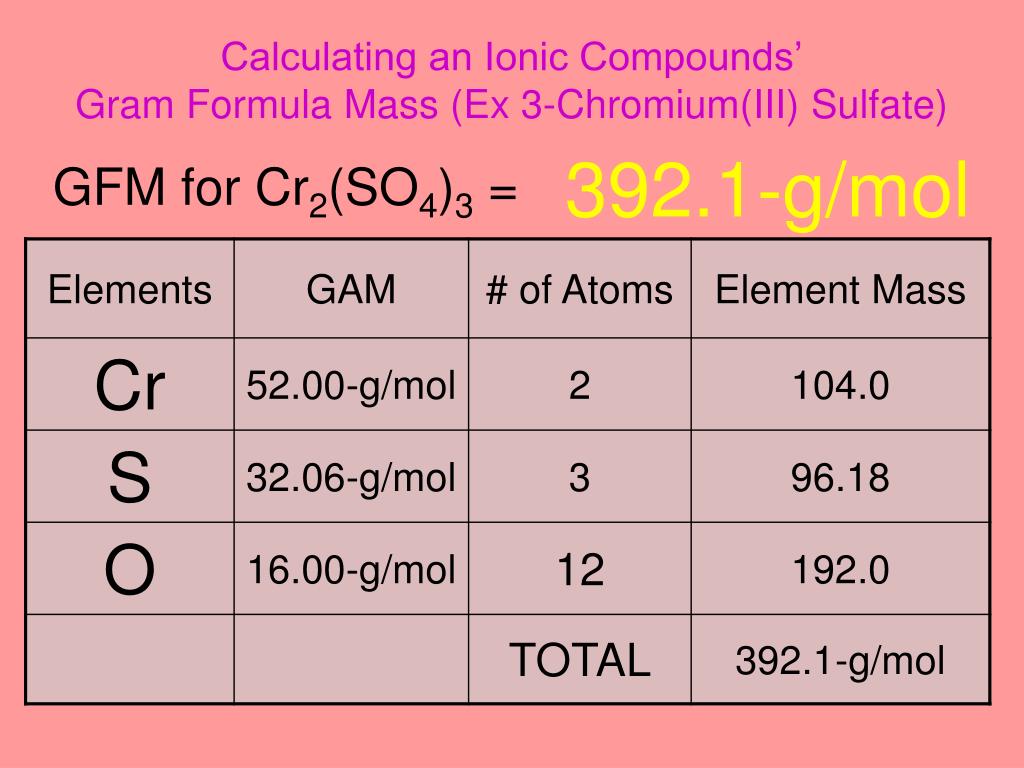
11. What is the gram formula mass of chromic sulfate, Cr2(SO4)3?8.148.19C. 344.2 gD. 392.2912. What is - Brainly.ph
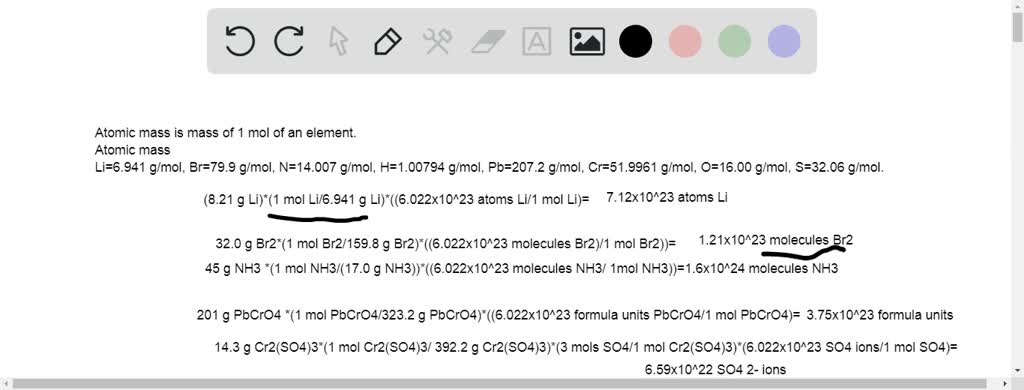
SOLVED:Calculate the following. number of atoms in 8.21 \mathrm{~g} \mathrm{Li} number of atoms in 32.0 \mathrm{~g} \mathrm{Br}_{2} number of molecules in 45 \mathrm{~g} \mathrm{NH}_{3} number of formula units in 201 \mathrm{~g} \mathrm{PbCrO}_{4}
Balance the following equations by oxidation number method 1. K2Cr2O7 + KI + H2SO2 → K2SO4 + Cr2(SO4)3 + I2 + H2O - Sarthaks eConnect | Largest Online Education Community

Calculate the equivalent weight of Cr2(So4)3. In given reaction Cr2(So4)3 + H2o2+NaOH Na2CrO4 + - Brainly.in